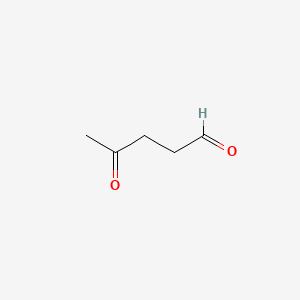
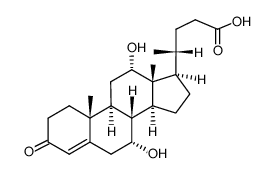
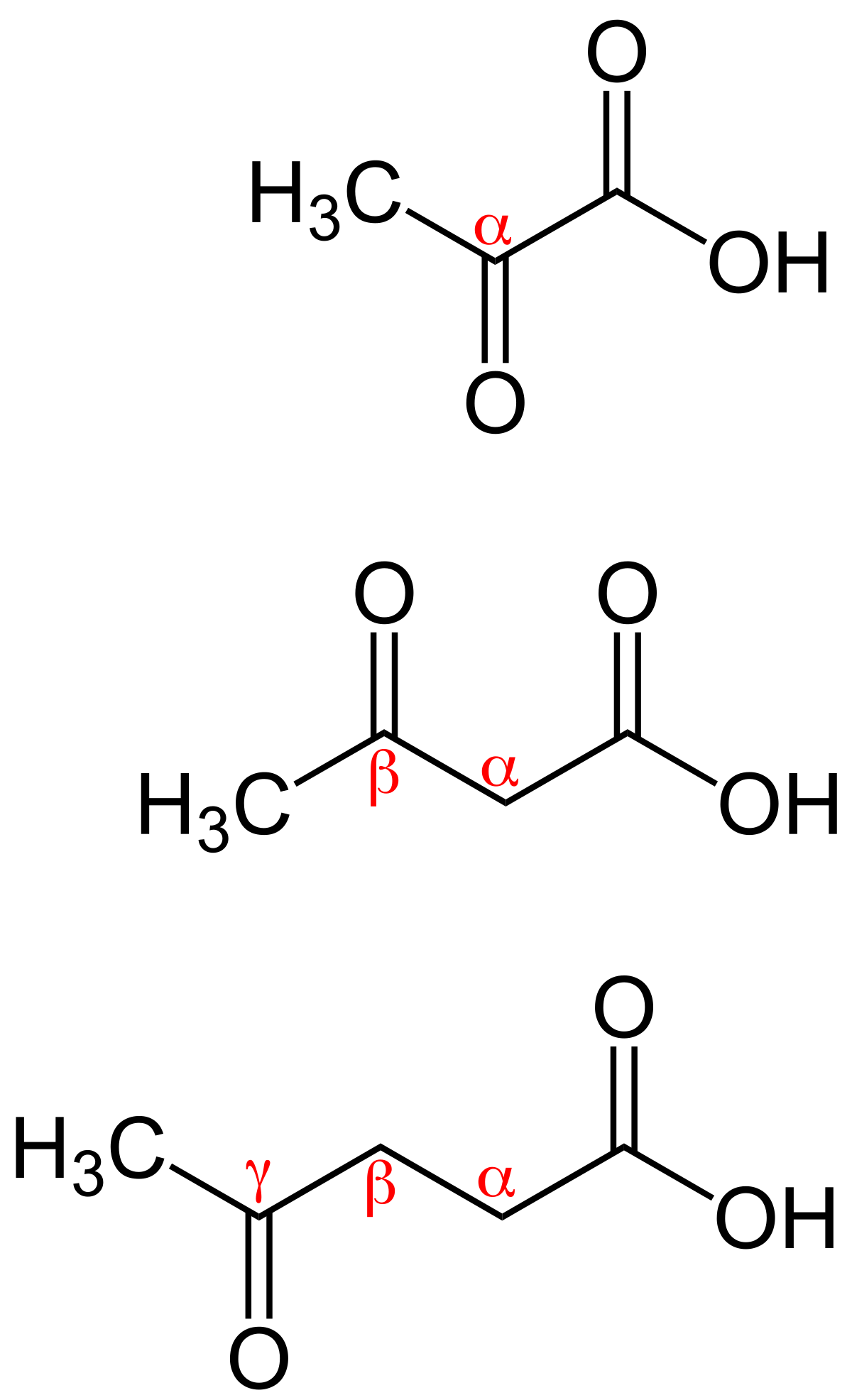
Reaction of X and phenylacetic acid forms an intermediate Y, which undergoes an intramolecular reaction to yield rofecoxib. Rofecoxib is a nonsteroidal anti-inflammatory agent once marketed under the trade name Vioxx, now

Draw the electron-pushing mechanism for the propagation steps of the allylic bromination reaction below. You may omit NBS in your mechanism and use Br-cdot and Br2. | Homework.Study.com

PDF) Stereochemistry. XXXIV. Nitro compounds. 7. Chemistry of 3-oxo-2-phenylindolenine | Alfred Hassner - Academia.edu
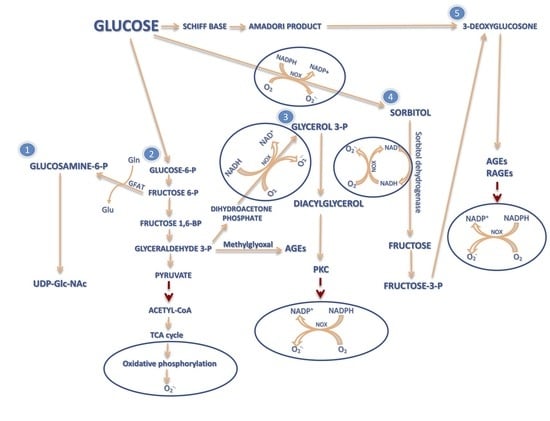
Molecules | Free Full-Text | 8-Oxo-7,8-Dihydro-2′-Deoxyguanosine (8-oxodG) and 8-Hydroxy-2′-Deoxyguanosine (8-OHdG) as a Potential Biomarker for Gestational Diabetes Mellitus (GDM) Development

PDF) Structure–function characterization of an aldo–keto reductase involved in detoxification of the mycotoxin, deoxynivalenol

P450 inhibitor ketoconazole increased the intratumor drug levels and antitumor activity of fenretinide in human neuroblastoma xenograft models - Lopez‐Barcons - 2017 - International Journal of Cancer - Wiley Online Library