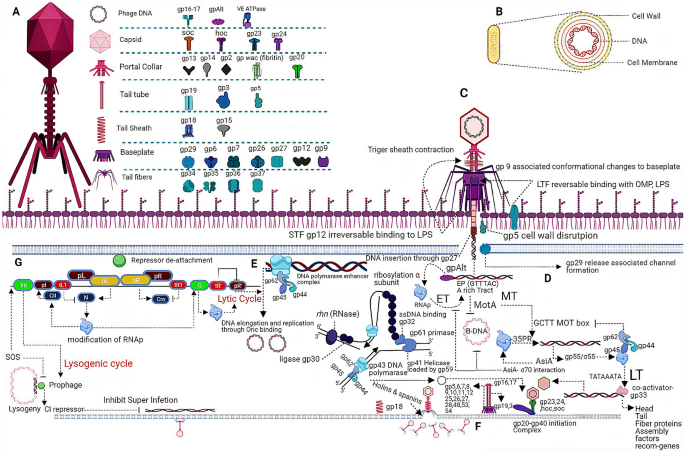
Characterization of the interactions between Escherichia coli receptors, LPS and OmpC, and bacteriophage T4 long tail fibers - Washizaki - 2016 - MicrobiologyOpen - Wiley Online Library

Receptors of Salmonella phages. Phages can use a number of cell surface... | Download Scientific Diagram

Interactions of bacteriophage T4 adhesin with selected lipopolysaccharides studied using atomic force microscopy | Scientific Reports

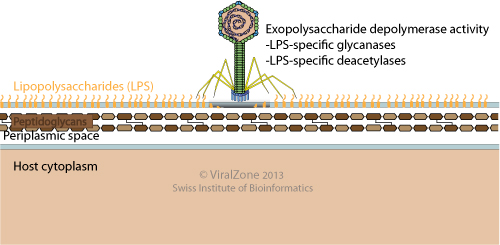
Not a barrier but a key: How bacteriophages exploit host's O‐antigen as an essential receptor to initiate infection - Broeker - 2017 - Molecular Microbiology - Wiley Online Library
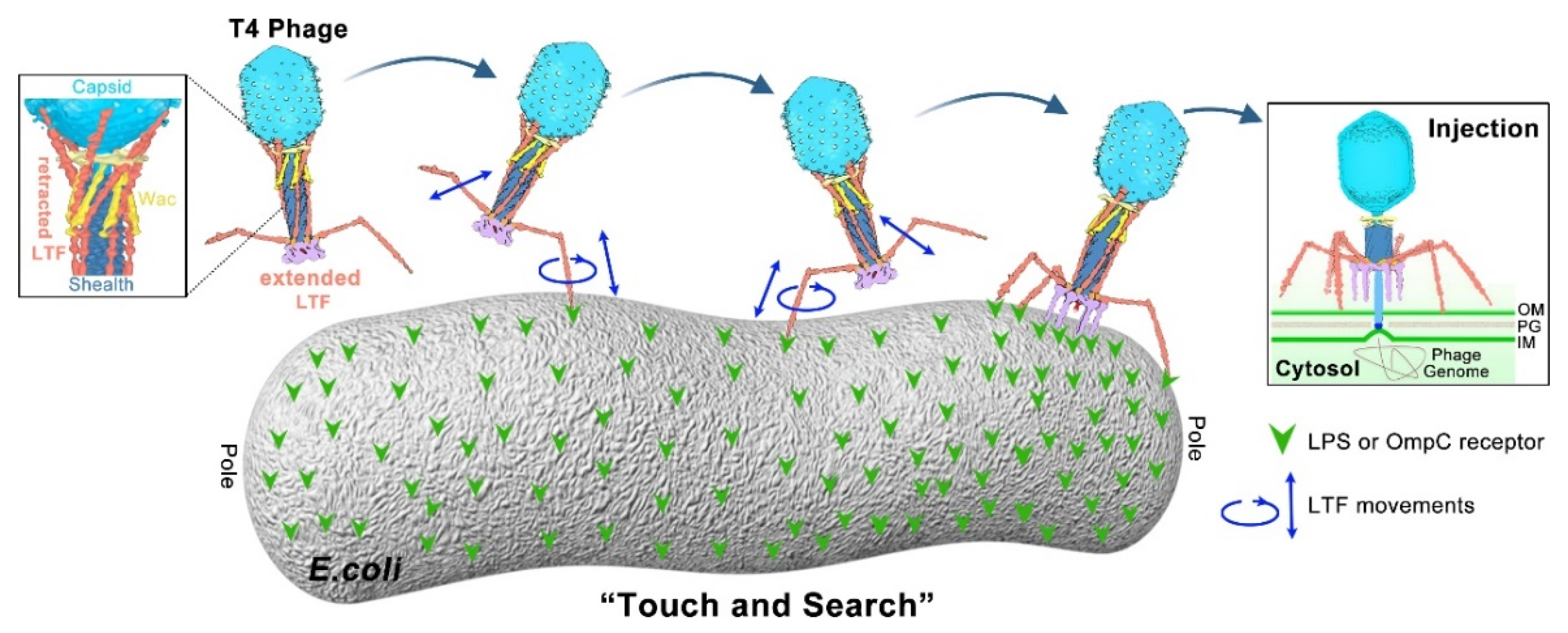
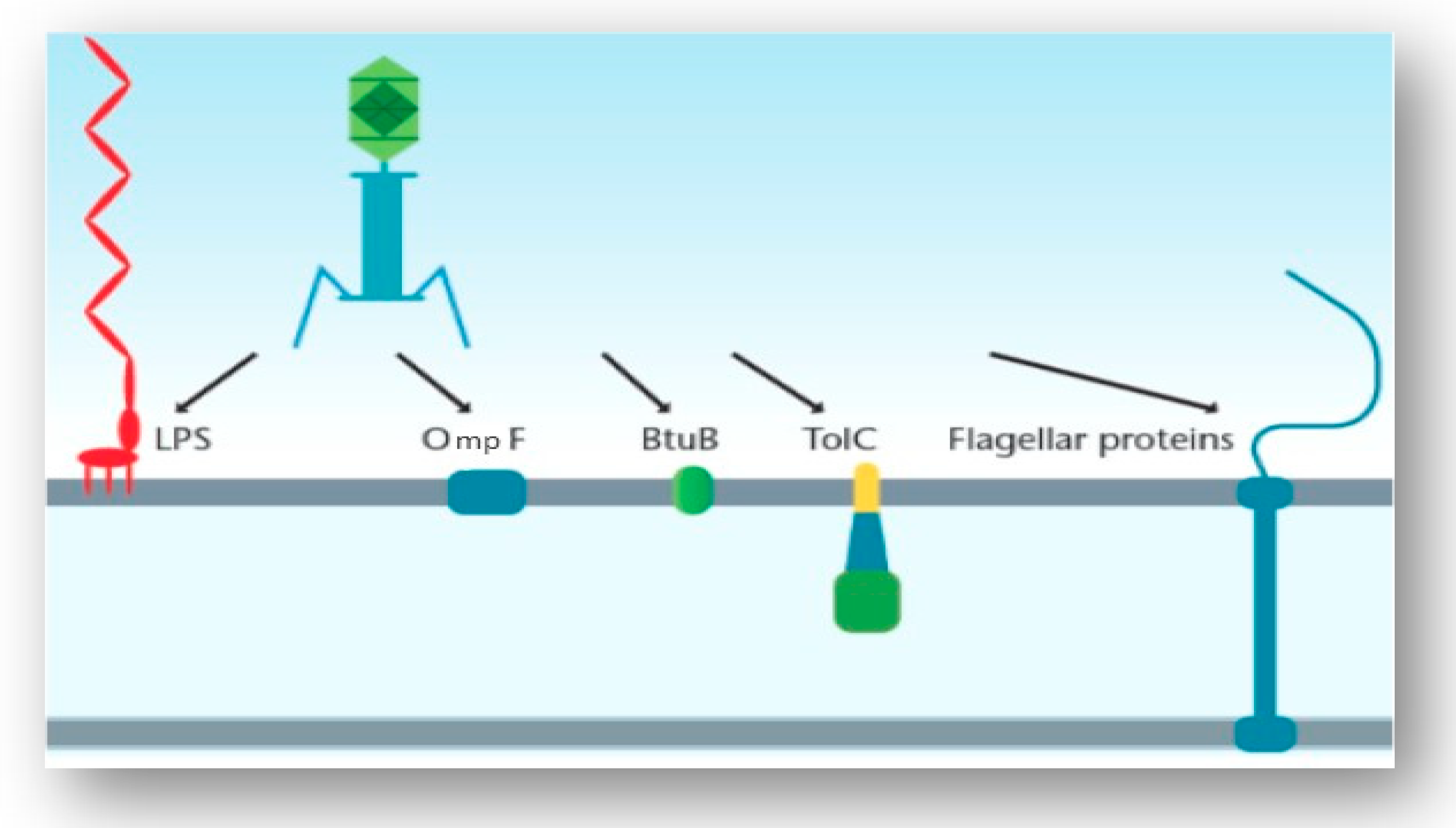
IJMS | Free Full-Text | Understanding Bacteriophage Tail Fiber Interaction with Host Surface Receptor: The Key “Blueprint” for Reprogramming Phage Host Range

Systematic exploration of Escherichia coli phage-host interactions with the BASEL phage collection | bioRxiv
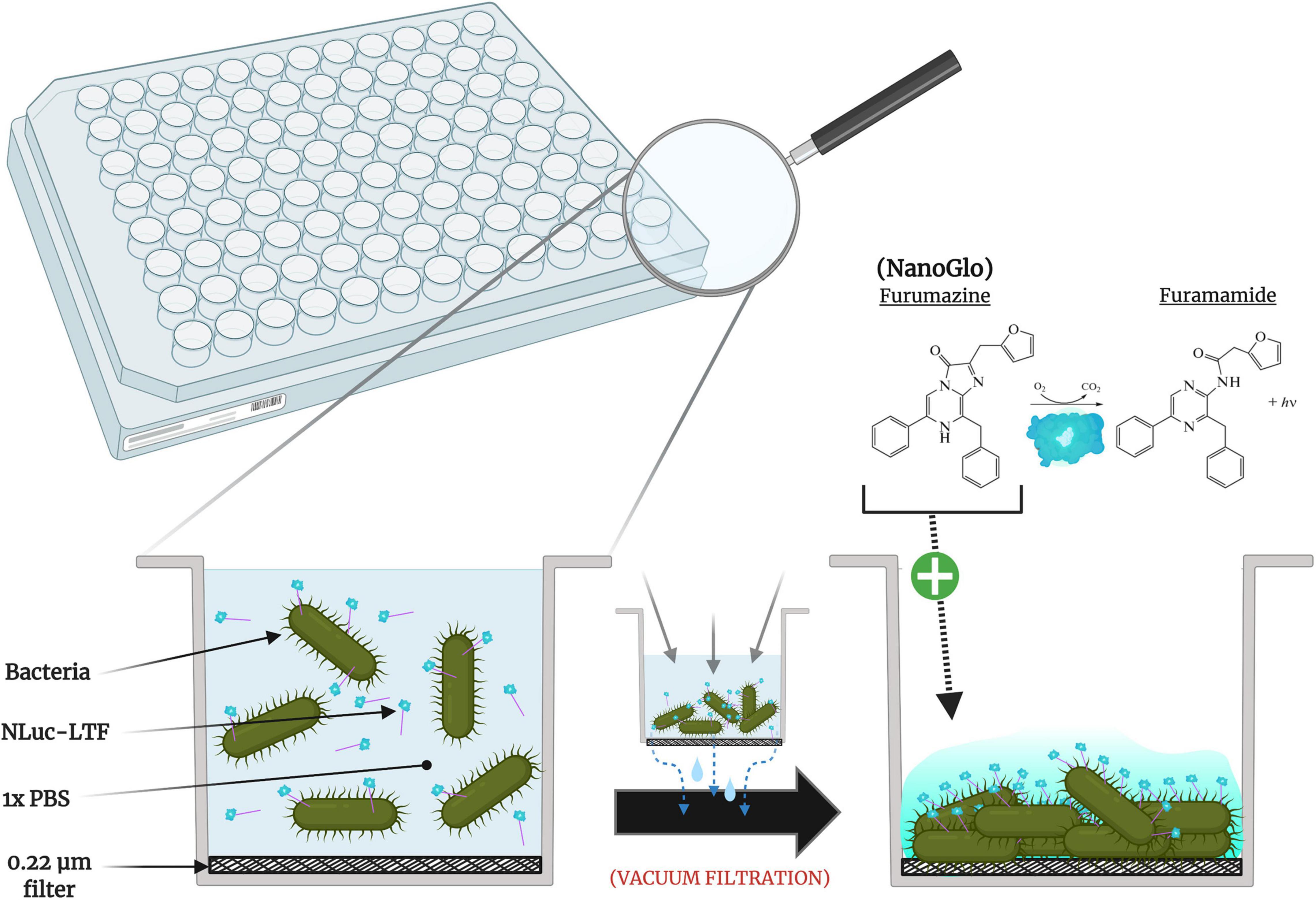
Frontiers | Evaluating Phage Tail Fiber Receptor-Binding Proteins Using a Luminescent Flow-Through 96-Well Plate Assay
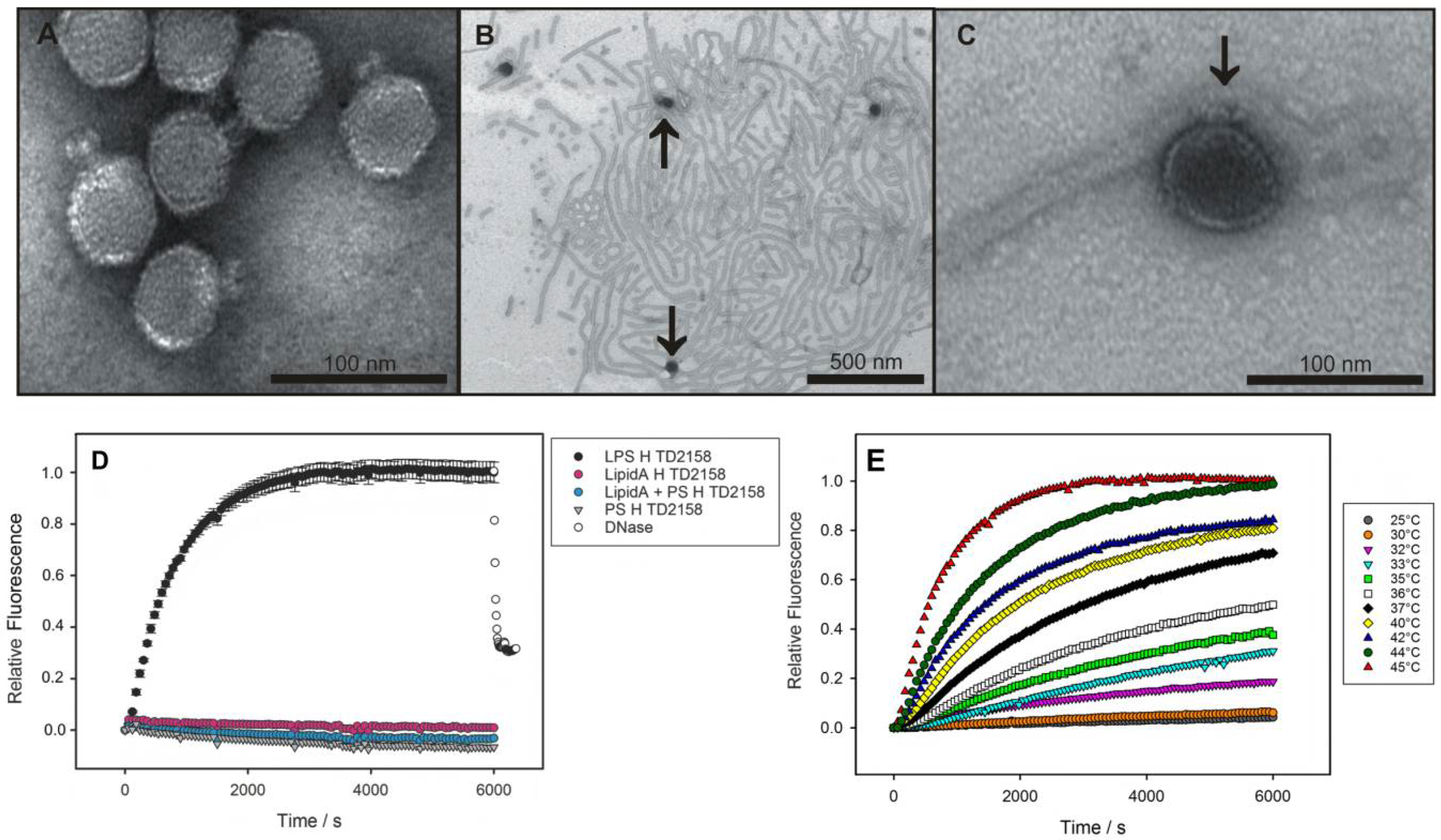
Viruses | Free Full-Text | In Vitro Studies of Lipopolysaccharide-Mediated DNA Release of Podovirus HK620

Systematic Discovery of Salmonella Phage-Host Interactions via High-Throughput Genome-Wide Screens | bioRxiv
Receptor Diversity and Host Interaction of Bacteriophages Infecting Salmonella enterica Serovar Typhimurium | PLOS ONE

Novel Host Recognition Mechanism of the K1 Capsule-Specific Phage of Escherichia coli: Capsular Polysaccharide as the First Receptor and Lipopolysaccharide as the Secondary Receptor | Journal of Virology

More than Rotating Flagella: Lipopolysaccharide as a Secondary Receptor for Flagellotropic Phage 7-7-1 | Journal of Bacteriology

Novel Host Recognition Mechanism of the K1 Capsule-Specific Phage of Escherichia coli: Capsular Polysaccharide as the First Receptor and Lipopolysaccharide as the Secondary Receptor | Journal of Virology
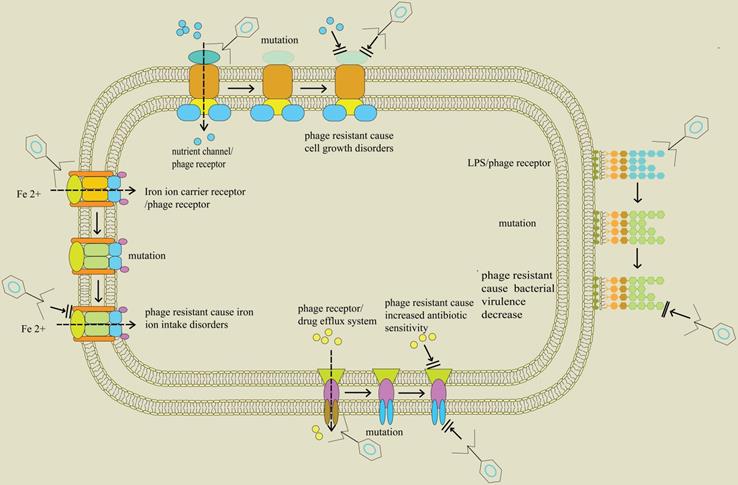
Fitness Trade-Offs Resulting from Bacteriophage Resistance Potentiate Synergistic Antibacterial Strategies

A lipopolysaccharide-dependent phage infects a pseudomonad phytopathogen and can evolve to evade phage resistance | bioRxiv

Phage–antibiotic combinations: a promising approach to constrain resistance evolution in bacteria - North - 2021 - Annals of the New York Academy of Sciences - Wiley Online Library