Fluorometric methods for determination of H2O2, glucose and cholesterol by using MnO2 nanosheets modified with 5-carboxyfluorescein | SpringerLink
![General Chemistry: Balancing Redox Reactions] is this "balance-able" or is there something wrong with the problem? : r/HomeworkHelp General Chemistry: Balancing Redox Reactions] is this "balance-able" or is there something wrong with the problem? : r/HomeworkHelp](https://preview.redd.it/gqgn3hnn3gp91.jpg?auto=webp&s=432f2eb7f0dc7f458e8a289268e284617ad3a4fa)
General Chemistry: Balancing Redox Reactions] is this "balance-able" or is there something wrong with the problem? : r/HomeworkHelp

In the following reaction: SO2(g) + 2H2S(g)→ 3S(s) + 2H2O(l) , the number of moles sulphur formed by 2 moles each of SO2 and H2S is :

Does the reaction rate depend on the concentration of the catalyst? For example, I found that the decomposition of H2O2 stops if we add excess MnO2. Are there any other examples like
Thermocatalytic Behavior of Manganese (IV) Oxide as Nanoporous Material on the Dissociation of a Gas Mixture Containing Hydrogen

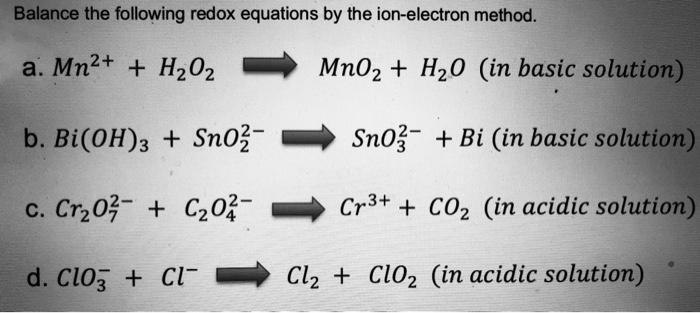
SOLVED: Mn2+ + H2O2 → MnO2 + H2O in basic medium Separate the reaction into two half-reactions and balance each of them.
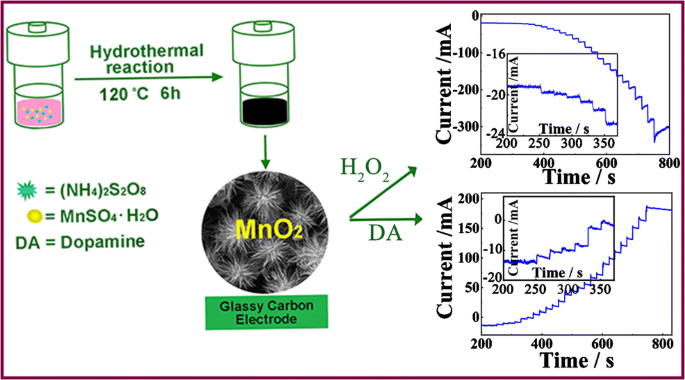
A hollow urchin-like α-MnO2 as an electrochemical sensor for hydrogen peroxide and dopamine with high selectivity and sensitivity | SpringerLink
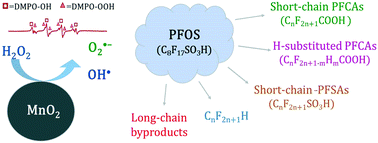
Degradation of PFOS by a MnO2/H2O2 process - Environmental Science: Water Research & Technology (RSC Publishing)

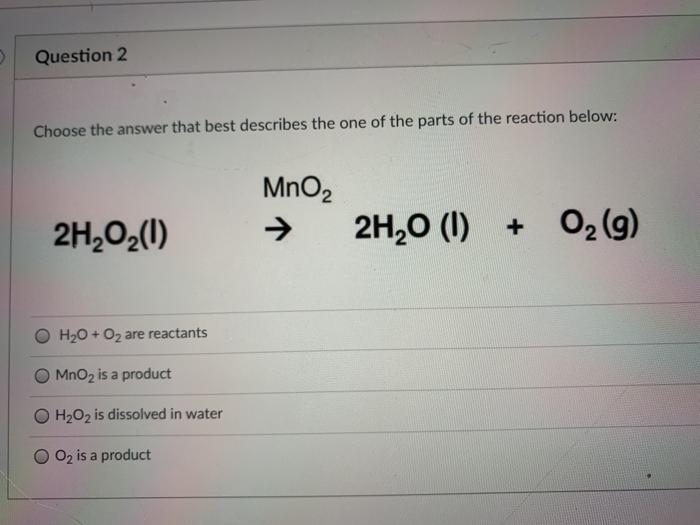
Question Video: Identifying the Correct Statement For the Decomposition of Hydrogen Peroxide Using a Manganese Dioxide Catalyst | Nagwa

SOLVED: Question 6 Not yet answered Marked out of 3 Flag question Balance the following redox reaction in basic aqueous solution: Mn2+ H2O2 MnOz H2O Select one: a. Mn2+ H2O2 SM 2

Study of photocatalytic decomposition of hydrogen peroxide over ramsdellite- MnO2 by O2-pressure monitoring - ScienceDirect

inorganic chemistry - Reaction intermediates of MnO2 catalyzed H2O2 decomposition reaction - Chemistry Stack Exchange