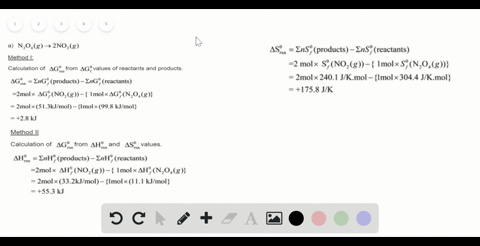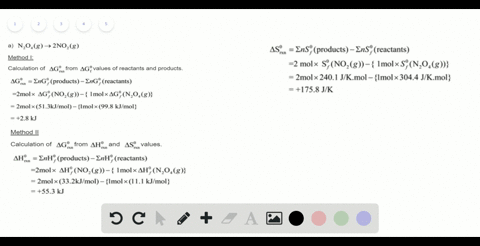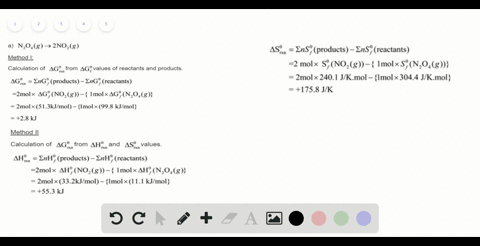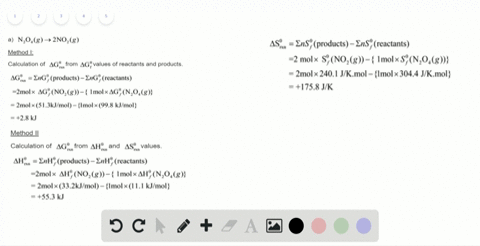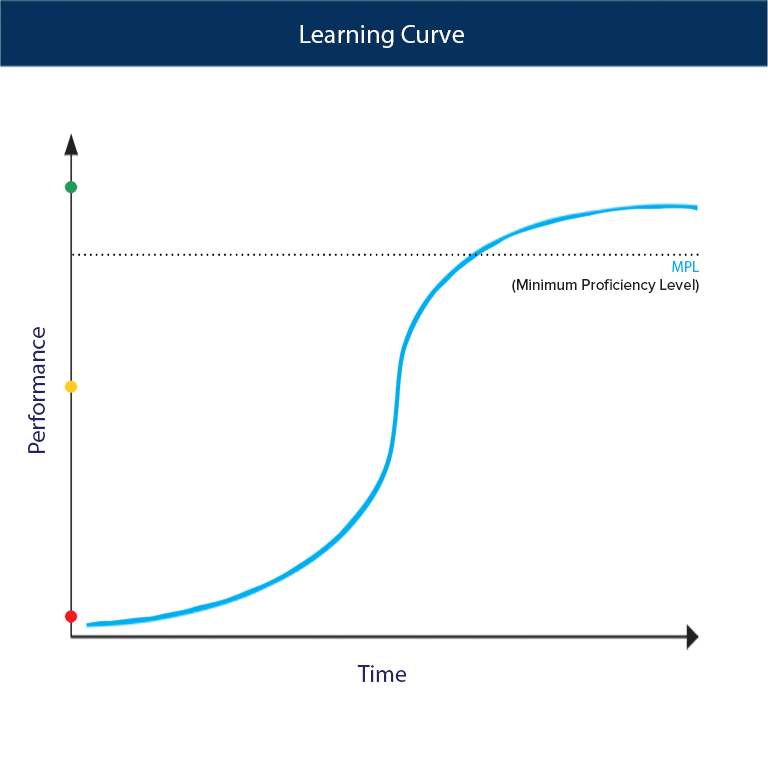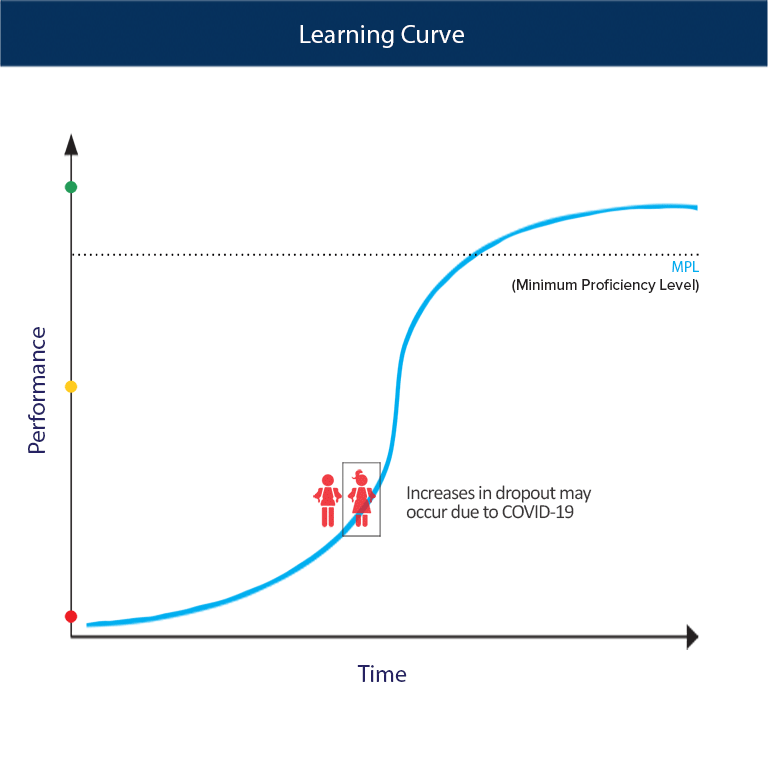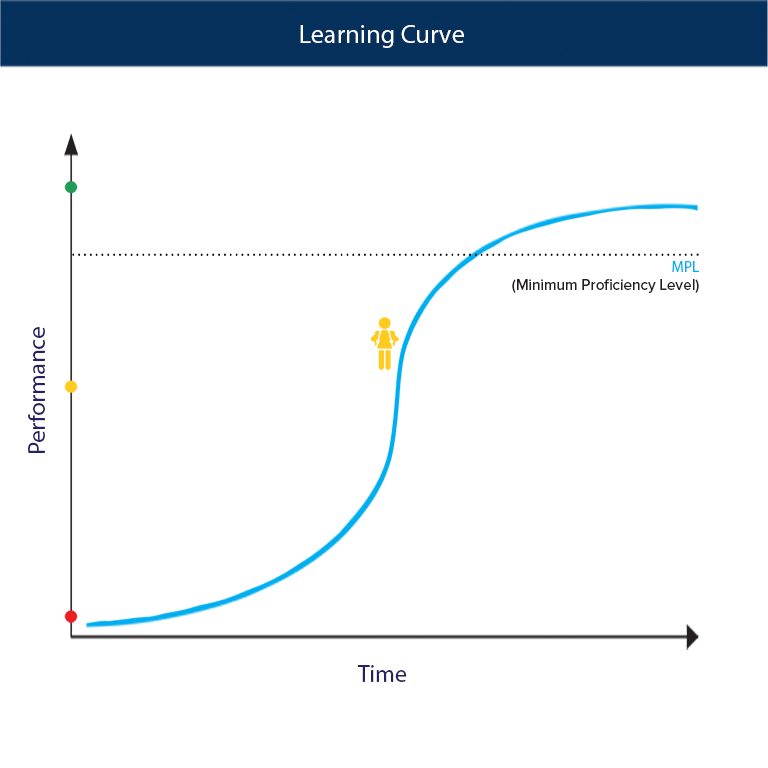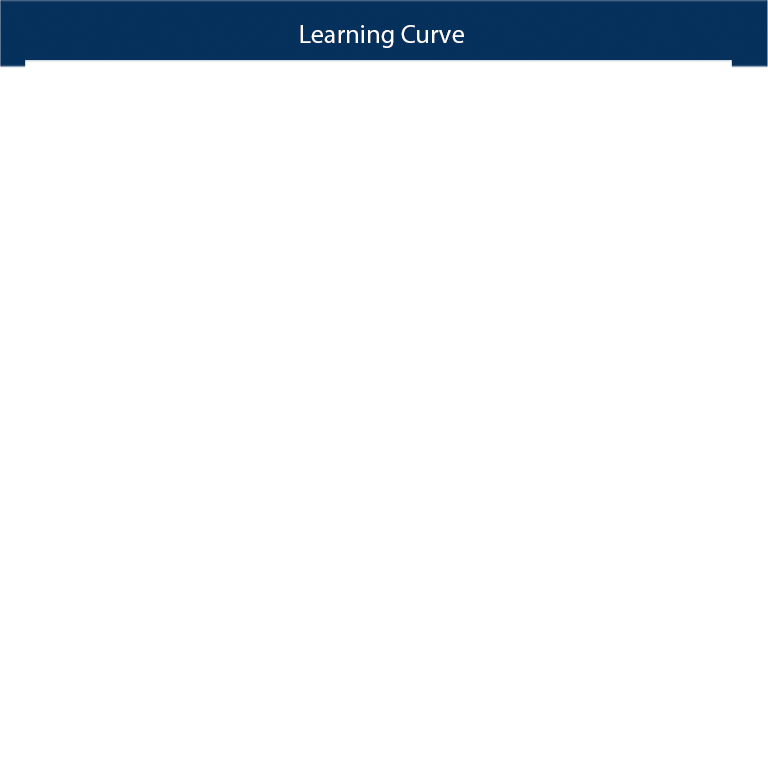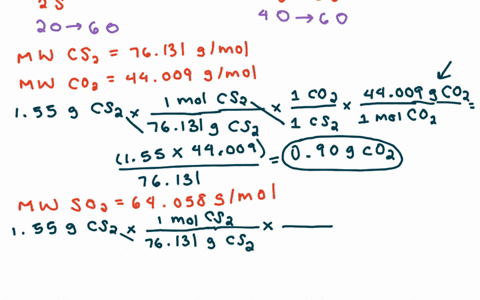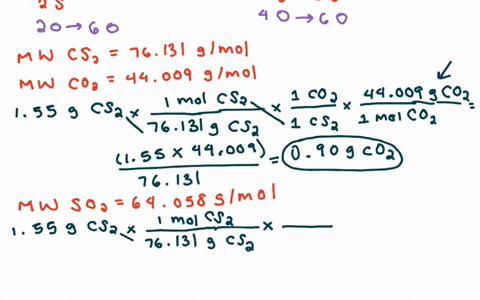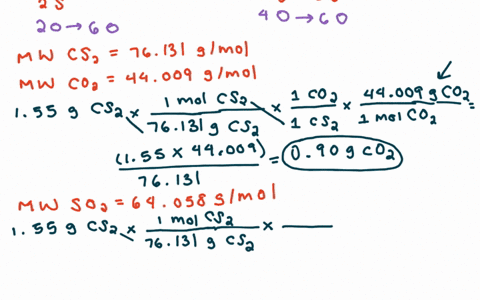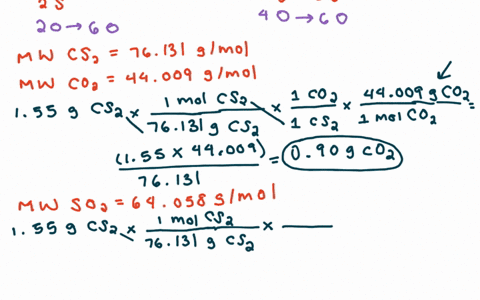
SOLVED:For each of the following unbalanced equations, calculate the mass of each product that could be produced by complete reaction of 1.55 g of the reactant indicated in boldface. a. 𝐂 𝐒2(l)+O2(g)
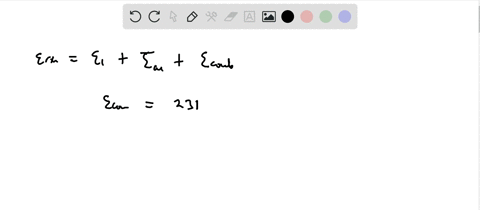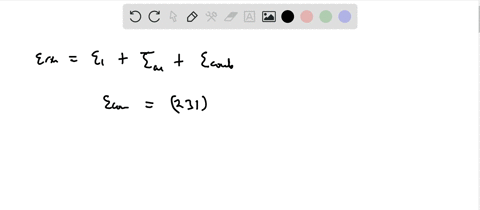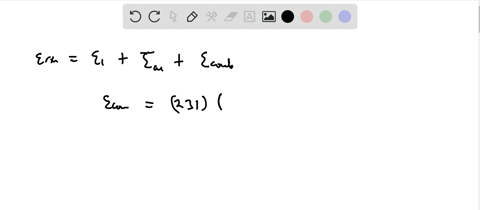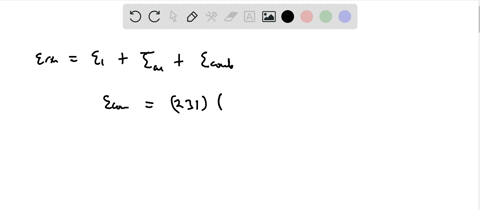
SOLVED:Calculate the value of the energy released (in aJ per ion pair) in the reaction described by the equation K(g)+Br(g) →K^+ Br^-(g) The first ionization energy of a potassium atom is 0.696