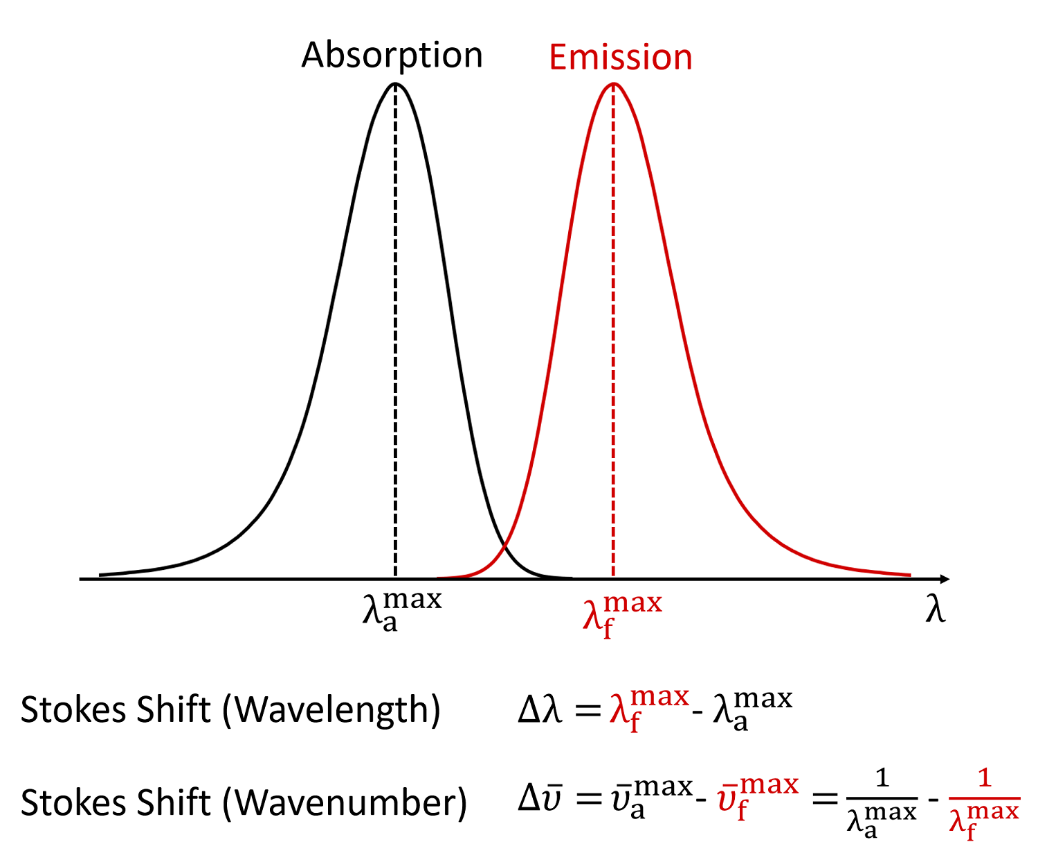
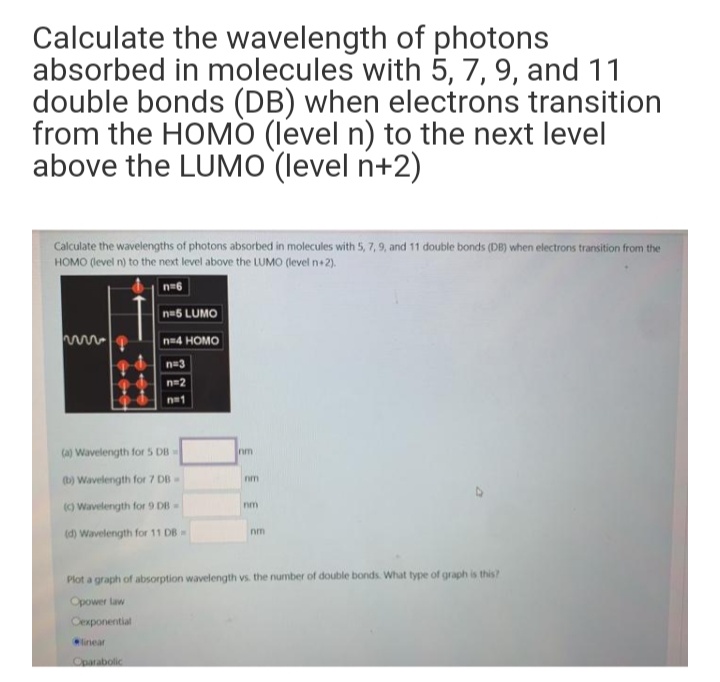
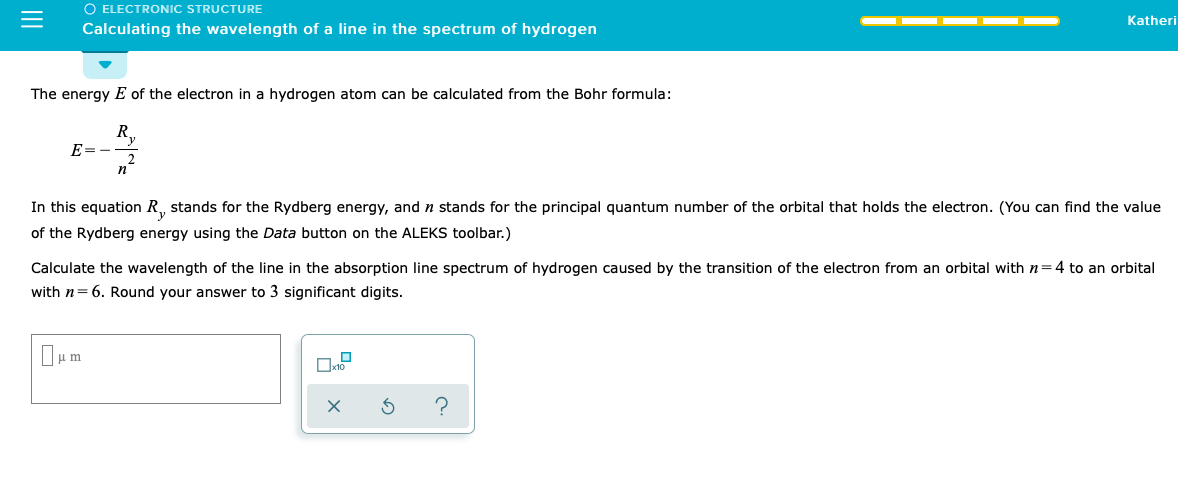
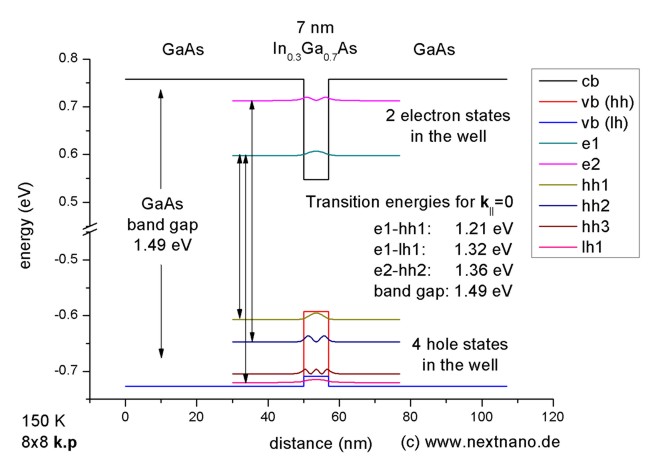
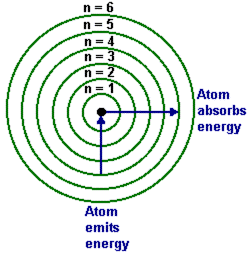
The longest wavelength doublet absorption transition is observed at 589 and 589.6 nm. Calculate the frequency of each transition and energy difference between two excited states.
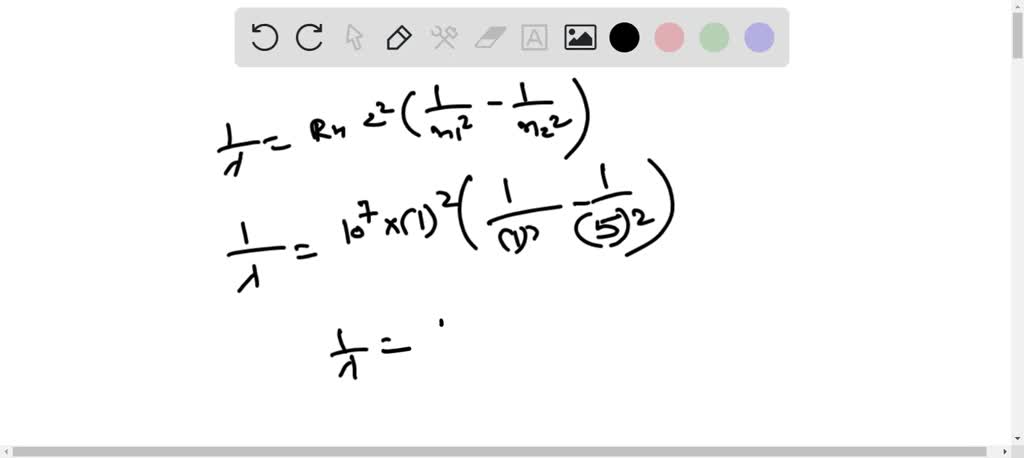
SOLVED: Calculate the wavelength of the line in the absorption line spectrum of hydrogen caused by the transition of the electron from an orbital with n=1 to an orbital with n=5. Round

In a one-body picture, there is only one transition in the absorption... | Download Scientific Diagram

The longest wavelength doublet absorption transition is observed at 589 and 589.6 nm. Calculate the frequency of each transition and energy difference between two excited states.

CHEM1011 notes for 8 topics - Atomic spectra & Rydberg 1. Relate the lines and series in the - Studocu
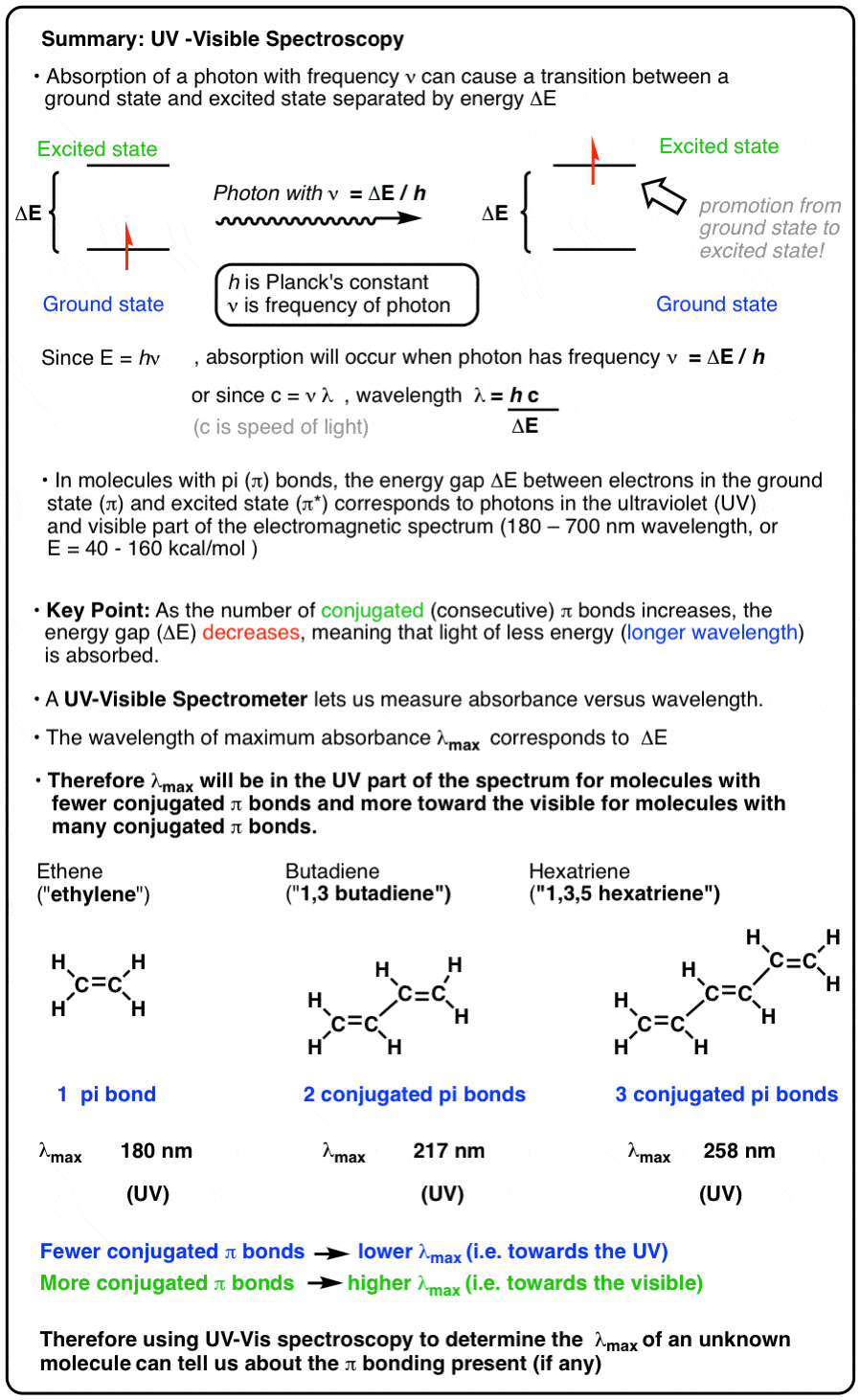
CALCULATION OF THE TRANSITION DIPOLE MOMENT OF THE ˜A ← ˜X ELECTRONIC TRANSITION OF THE C2H5O2 FROM THE PEAK ABSORPTION CROS

Femtosecond Absorption Spectroscopy of Transition Metal Charge-Transfer Complexes | Accounts of Chemical Research
CALCULATION OF THE TRANSITION DIPOLE MOMENT OF THE $\tilde{A}\leftarrow \tilde{X}$ ELECTRONIC TRANSITION OF THE C$_2$H$_5$O$_2$ FROM THE PEAK ABSORPTION CROSS-SECTION
![PDF] Calculation of single-beam two-photon absorption transition rate of rare-earth ions using effective operator and diagrammatic representation | Semantic Scholar PDF] Calculation of single-beam two-photon absorption transition rate of rare-earth ions using effective operator and diagrammatic representation | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/b0045c9a6c975d5467e09bdc57a57de9752d40b5/8-Figure1-1.png)
PDF] Calculation of single-beam two-photon absorption transition rate of rare-earth ions using effective operator and diagrammatic representation | Semantic Scholar

Transition cross-sections, explained by RP Photonics Encyclopedia; laser, emission, absorption cross-sections, gain coefficient