Brain Tumour Magazine: World Edition 2018/2019 by The International Brain Tumour Alliance (IBTA) - Issuu
/cloudfront-eu-central-1.images.arcpublishing.com/prisa/OGT2FFIPXFF6NE3IF5O7QIK6N4.jpg)
European Medicines Agency warns of deaths due to prolonged use of drugs combining codeine and ibuprofen | Science & Tech | EL PAÍS English Edition
Open Letter to J&J: Calling for affordable access to critical TB drug bedaquiline | Médecins Sans Frontières Access Campaign
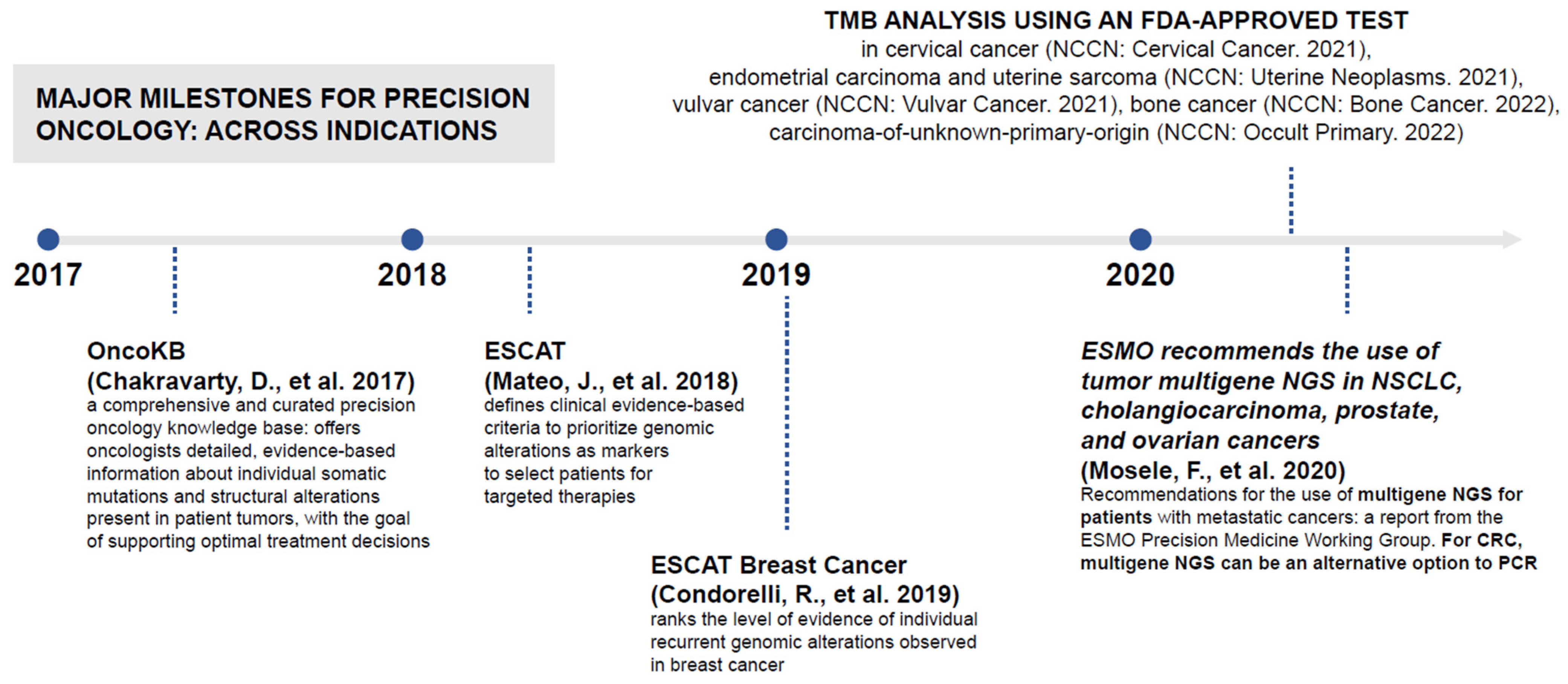
JPM | Free Full-Text | Identifying the Steps Required to Effectively Implement Next-Generation Sequencing in Oncology at a National Level in Europe

Public consultation on key principles for the electronic product information of EU medicines | European Medicines Agency
European Medicines Agency pre-authorisation procedural advice for users of the centralised procedure