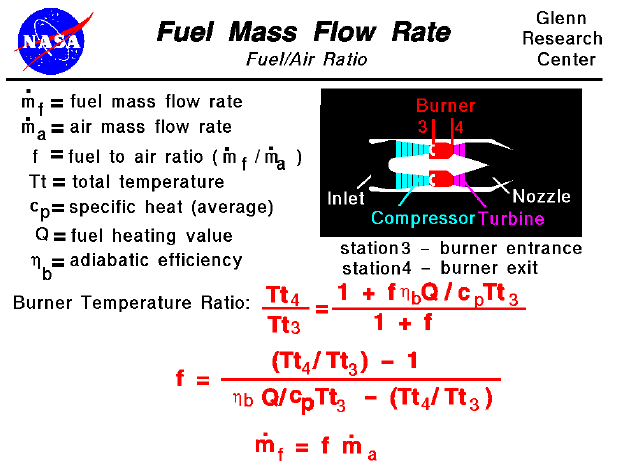
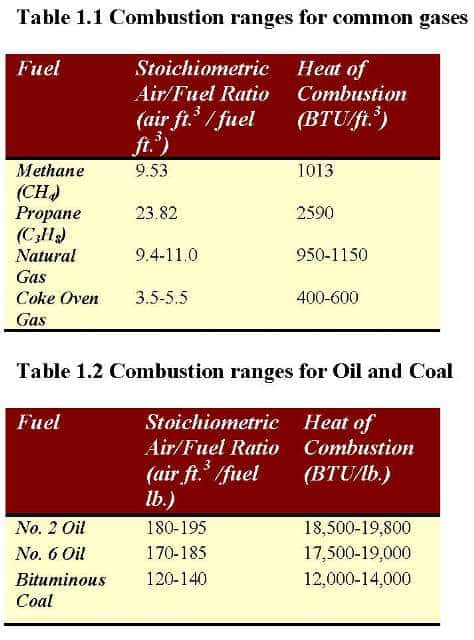
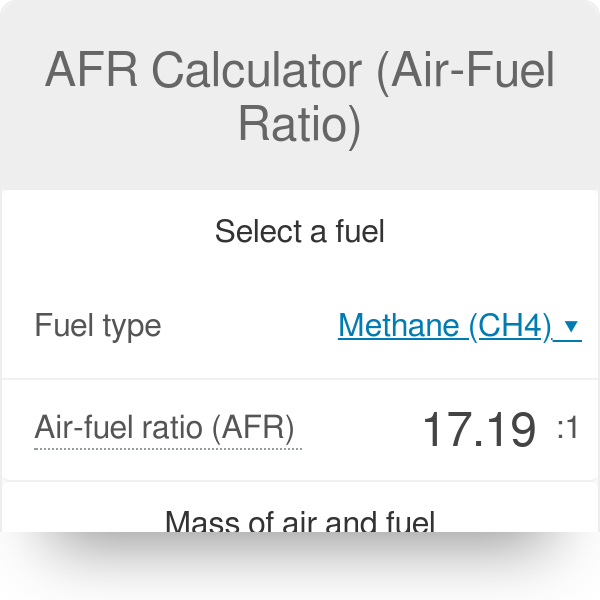
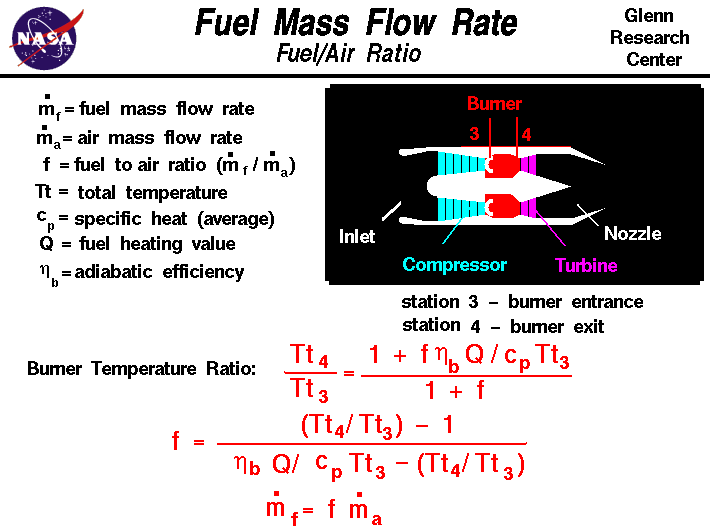
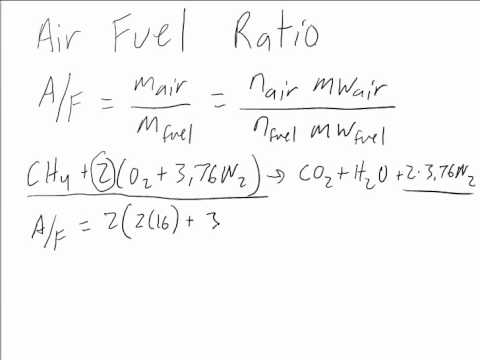How would you balance a combustion reaction like this and how do you calculate the air/fuel ratio? : r/chemistryhelp

SOLVED: 'Example 3: Methanol is burned with 20% excess air the sticometreic air-fuel ratio and the actual air-fuel ratio and equivalence ratio if air supplied at bar and 27 %C. Calculate the
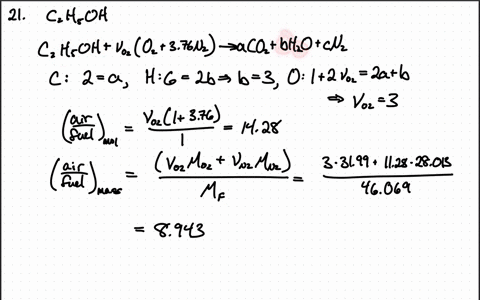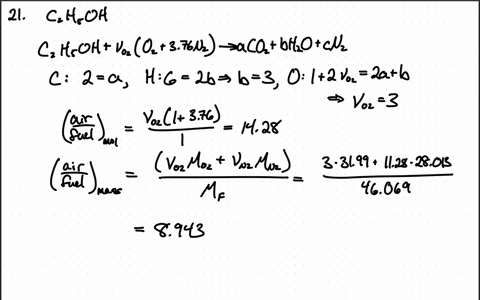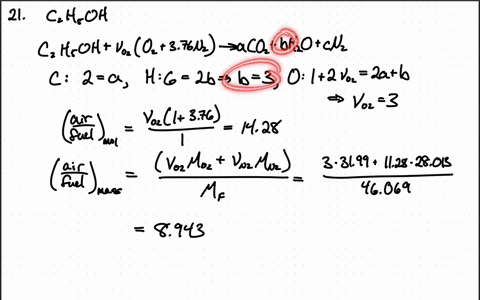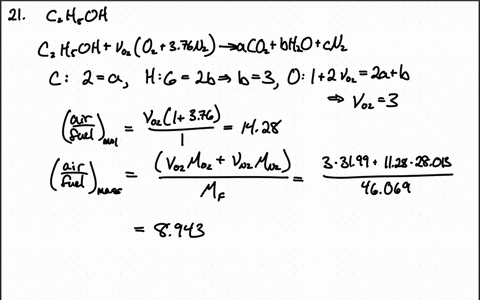
SOLVED:Calculate the theoretical air-fuel ratio on a mass and mole basis for the combustion of ethanol, C2 H5 OH.